Listen to the article here:
The liver is an amazing and necessary organ. It can regenerate from a minor injury, break down dangerous chemicals and drugs, and help maintain the proper balance of nutrients, fats, and sugars in the body. It has hundreds of other important roles as well. With all this responsibility, we are in trouble when the liver stops working.
One way the liver stops working is through non-alcoholic steatohepatitis (NASH). Steato- means fat, hepato- indicates the liver, and -itis means inflammation. Steatohepatitis is inflammation of the liver caused by fat accumulation. This is a progressive form of non-alcoholic fatty liver disease – the most common liver disorder in western countries. Progressive means that this disease gets more severe over time.
NASH is a large problem in America, affecting 3-12% of adults. Furthermore, NASH can lead to cirrhosis, where the liver is permanently damaged, and lead to possible liver transplantation. Over a million adults in America have NASH-related cirrhosis.
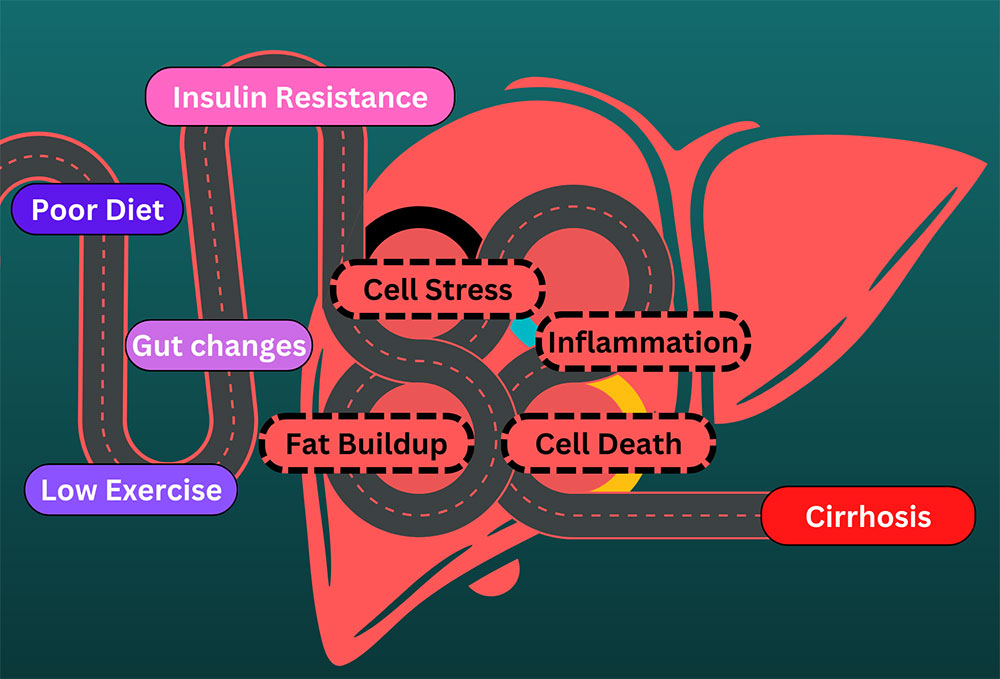
How do we get NASH? As the name indicates, this is not caused by alcohol. There are many pathways to developing NASH, but the underlying cause may be excess carbs and fatty acids. This can be due to diet or behavior, underlying genetics, or associated syndromes. Some of the syndromes associated with NASH are:
- Metabolic syndrome
- Obesity
- Type 2 diabetes
- High blood pressure
- Dyslipidemia
- Hypothyroidism
- Cardiovascular risk
- Advanced liver problems
The underlying mechanism of NASH can be very complex. A leading precursor to NASH is insulin resistance, where cells fail to respond to insulin. Conditions that cause or are caused by insulin resistance, such as type II diabetes and metabolic syndrome, may increase your chances of developing NASH. They also may develop or worsen as NASH symptoms get worse.
Insulin resistance causes different types of fats to accumulate in the liver. This makes it very difficult for the liver to process the fats and they ultimately build up in liver cells. These fats, especially ones called nonesterified fatty acids (NFEAs), are very dangerous. They cause damage to liver cells and can also activate cytokines that start the inflammation process.
Eventually, we get NASH, an inflammation cascade in the liver caused by fats. Cytokines start inflammation in the liver. Cell death attracts the immune system, which enters and causes inflammation while trying to help. Liver cells die and are less able to process fats, which leads to a compounding effect. Eventually, we may transition to cirrhosis, where permanent liver scarring and damage occur.
There are few treatments on the market for NASH. As usual, the primary therapy for NASH is a good diet and regular exercise. Medicinal remedies are all in the experimental phases. Potential targets include increasing insulin sensitivity, decreasing fat creation, decreasing circulating fats, breaking fats down, and anti-inflammation treatments.
One way to decrease circulating fats is by expelling them through the digestive system. In the liver, moving fats to the gut is regulated by thyroid hormones. Thyroid hormones activate receptors, which exist all over the body and cause many different effects. Thyroid hormone receptor Beta (THR-β) exists almost exclusively in the liver. Scientists are working to create medicines that activate THR-β and help clear fats from the liver in NASH patients. If you are interested in participating in a research study, contact your local ENCORE Research Group site today!
Sources:
Noureddin, M., & Sanyal, A. J. (2018). Pathogenesis of NASH: the impact of multiple pathways. Current Hepatology Reports, 17, 350-360.
Parthasarathy, G., Revelo, X., & Malhi, H. (2020). Pathogenesis of nonalcoholic steatohepatitis: an overview. Hepatology communications, 4(4), 478-492. https://doi.org/10.1002/hep4.1479
Pierantonelli, I., & Svegliati-Baroni, G. (2019). Nonalcoholic fatty liver disease: basic pathogenetic mechanisms in the progression from NAFLD to NASH. Transplantation, 103(1), e1-e13. . https://doi.org/10.1097/TP.0000000000002480
Pramfalk, C., Pedrelli, M., & Parini, P. (2011). Role of thyroid receptor β in lipid metabolism. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1812(8), 929-937. https://doi.org/10.1016/j.bbadis.2010.12.019

