I remember the early days of COVID-19, everything was new and scary and dangerous and no one knew what was going on. It seemed to be very dangerous for two groups of people, those of advanced age and those who were immunocompromised. Vaccines rolled out, testing became easy, and things have calmed down quite a bit. But corona isn’t all limes on the beach, as any virus, it’s still dangerous for people who are immunocompromised. What does this term mean, who are the immunocompromised, and is there anything they can do in the new reality of COVID-19? Immunocompromised is a broad term. It indicates that a person’s immune system cannot generate an appropriate response to infection. When bacteria or viruses make it inside these people’s bodies, they cause much more damage and are very hard to control. The immune system is very, very complicated, so there are many ways a person can become immunocompromised. We can generally lump these into two categories: primary and secondary immunodeficiency. Primary immunodeficiency means the condition is built-in to the body; it’s genetic. Primary immunodeficiencies are fairly rare, with <0.1% of the population experiencing them. The rest of the almost 3% of people who have immunodeficiencies have secondary immunodeficiencies.Infectious diseases (such as HIV), malnutrition, age, surgery, environmental stress, and immunosuppressive drugs can all cause secondary immunodeficiency. Immunodeficiency affects millions of Americans. Women are twice as likely as men to have immunodeficiency; it is most common in white Americans and those aged 50-59. Nearly 3% of the population – over 9 million Americans – are suspected to have immunodeficiency. Unfortunately, immunodeficiency can greatly reduce a person’s ability to deal with a COVID-19 infection. The most obvious problem is that immunocompromised people are more susceptible to severe symptoms. A disproportionate amount of people who are hospitalized for COVID-19 are immunocompromised. Immunodeficiency doesn’t compromise, it packs a double-whammy. Those with a weak immune system also find vaccines less effective! In fact, 44% of people who had “breakthrough” cases (where they were vaccinated but still hospitalized) were immunocompromised. This is because the body is unable to produce enough protective antibodies for the body to be protected – a process called seroconversion. Seroconversion is when antibodies are able to be detected in the blood. With vaccines, successful seroconversion indicates that the body is protected and has the equipment necessary to put up a good fight against the COVID-19 virus. When vaccinated against COVID-19, people with healthy immune systems showed seroconversion rates of 99%. The type of immunocompromisation affects how well vaccines produce seroconversion. People with solid tumor cancers, such as breast, colon, prostate, and lung cancer show seroconversion rates of 92%. Immune-inflammatory disorders like lupus, primary biliary cholangitis, psoriasis, and rheumatoid arthritis have seroconversion rates reduced to 78%. Vaccine effectiveness in people with blood cancers such as lymphomas, myeloma, and leukemia drops to 64%. Those with organ transplants have to be on strong immunosuppressive drugs to avoid organ rejection and because of this they have the lowest rates of seroconversion, only 27%.
Some factors of immunocompromisation. Adapted from Chinen, J., & Shearer, W. T. (2010).
So what can immunocompromised people do to protect themselves against COVID-19? A lot of the same things as people who are immunocompetent! High quality masks and respirators can help. Avoiding crowds and indoor areas with poor ventilation is a must. Washing hands with soap and water is critical, though hand sanitizer is a good second option. Immunocompromised people who contract COVID-19 should contact their doctor or other healthcare provider right away. Isolating and using masks to prevent the spread is always a good idea. Immunocompromised people may also keep infections from getting out of control if their medical provider recommends an antiviral medication or convalescent plasma. Of course, the best way to avoid getting sick with COVID-19 is through prevention, including vaccines. A 27% seroconversion rate is much better than 0% after all. And there may be more hope for immunocompromised people, as new vaccines are being developed to serve this community. Staff Writer / Editor Benton Lowey-Ball, BS, BFA
References: Boyle, J. M., & Buckley, R. H. (2007). Population prevalence of diagnosed primary immunodeficiency diseases in the United States. Journal of clinical immunology, 27, 497-502. https://link.springer.com/article/10.1007/s10875-007-9103-1 Chinen, J., & Shearer, W. T. (2010). Secondary immunodeficiencies, including HIV infection. Journal of Allergy and Clinical Immunology, 125(2), S195-S203. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6151868/ Harpaz, R., Dahl, R. M., & Dooling, K. L. (2016). Prevalence of immunosuppression among US adults, 2013. Jama, 316(23), 2547-2548. https://jamanetwork.com/journals/jama/fullarticle/2572798 National Institute of Health. (July 21, 2023). Special considerations in people who are immunocompromised. COVID-19 Treatment Guides, https://www.covid19treatmentguidelines.nih.gov/special-populations/immunocompromised/ Parker, E. P., Desai, S., Marti, M., Nohynek, H., Kaslow, D. C., Kochhar, S., … & Wilder-Smith, A. (2022). Response to additional COVID-19 vaccine doses in people who are immunocompromised: a rapid review. The Lancet Global Health, 10(3), e326-e328. https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(21)00593-3/fulltext SY, L. A. W., SC, C. L. L., & Muthiah, L. M. (2021). Efficacy of COVID-19 vaccines in immunocompromised patients: A systematic review and meta-analysis. medRxiv. https://doi.org/10.1136/bmj-2021-068632
Scroll down to listen to this article.

Listen to the article here:
GRID VIEW
I remember a joyous time when the world was young, and we had nothing to fear or worry about except climate change, an artificial intelligence cascade, political strife, and nuclear war. But we didn’t have to worry about COVID, so it was pretty idyllic. Now, over three years later, the biggest worries of COVID are winding down. The WHO declared COVID-19 to no longer be a global health emergency on May 5, 2023, and most places have lifted or lessened restrictions put in place to stem the spread. People have stopped wearing masks, and life seems to be back to normal. But not for everyone. Long COVID has been described as COVID symptoms that last longer than 5 weeks and may last months or longer. It affects around ⅓ of COVID patients, including over 85% of those that had to go to the hospital with severe symptoms. So what is long COVID, who gets it, how does it work, and is there anything to be done about it? Long COVID, also called long haul COVID, post-COVID syndrome (PCS), or Post-acute sequelae of SARS-CoV-2 infection (PASC), is different from person to person but can be debilitating. During acute (normal) COVID infections, people experience trouble with breathing, joint pain, headache, fatigue, stomach problems, and loss of smell and taste. When I had COVID, my sense of taste was so poor that I started liking Limp Bizkit again. Long COVID symptoms are similar to those of acute COVID. Long-lasting and often crushing fatigue is the most common symptom. I’ve heard anecdotal stories of people who run out of energy just deciding what to eat during the day; it can be very intense. Other symptoms include muscle pain, cognitive impairment such as brain fog, headaches, anxiety, and more. Around 140 million Americans have had COVID at some point. A recent study found that long COVID was more common and severe in people infected before the 2021 Omicron variant. Several other risk factors have also been identified. The severity of an acute COVID infection plays a role, as does having 5 or more separate symptoms. These can be mediated by being up to date on COVID vaccines. Long COVID is almost twice as common in women, and the risk is also increased if you are over 50 years old. Other health issues can also affect your chances. Being overweight, having psychiatric disorders, having asthma, and being in “poor general health” are risk factors. Interestingly, having latent Epstein-Barr virus might also increase your chances of developing long COVID. So how does all of this work? In the acute stage of COVID, the virus spreads rapidly and tries to reproduce. It gains entry into cells using its spike protein to fool a receptor on our cells called ACE2. It then hijacks cell machinery to make copies fast. This may kill the infected cells but also brings in the immune system, which kills the invaders (if we’re lucky) and tends to cause some damage through inflammation. It is thought that long COVID occurs through many mechanisms. The immune system can be disrupted, other infections can take hold, we can experience chronic inflammation, and some body systems can be messed up. Even worse, sometimes organs can be damaged from the infection, and the virus might stick around for a while! The major organs affected in long COVID can be deduced by looking at the symptoms. Trouble breathing, lung impairment, and low breath capacity may result from chronic inflammation and clotting in the lining of the lungs. Chest pain, irregular heartbeats, heart palpitations, and postural orthostatic tachycardia syndrome (POTS, low blood volume when standing up) are caused by chronic inflammation and cell death in the cardiovascular system. The cardiovascular system has an abundance of ACE2 receptors, meaning it is targeted for direct infection by the COVID virus. Fatigue, trouble sleeping, loss of taste and smell, and cognitive impairment are due to problems with our central nervous system. COVID can cross into the brain and cause inflammation of support cells and clotting (possibly leading to stroke!), and may also affect the brain stem. Nervous system problems affect around ⅓ of people by six months after COVID. There can also be problems with the kidney and pancreas. So what can we do? Our best bet is to reduce the effects of COVID in the first place – or avoid it altogether. Staying up to date on COVID vaccines and boosters lowers both the severity of acute COVID and the risks of developing long COVID. Continuing to wear masks, washing hands frequently, and being careful around sick people can also help, and staying healthy with diet and exercise can give a leg up. Unfortunately, if you already have long COVID, there are no meds proven to cure it. Treating symptoms is our current best practice. Supplements may help, including B vitamins, iron, magnesium, zinc, and selenium. Multivitamins, mineral supplements, and probiotics have shown preliminary promise, as has the antiviral paxlovid. ANY alteration of medication, including supplements, should be run by your doctor first to ensure they are safe and don’t interact with other conditions or medications you may be on. Non-pharmaceutical solutions may also help. Physical rehabilitation – including pulmonary rehabilitation – as well as mental health and social assistance are vital to making it down the long road of long COVID. Staff Writer / Editor Benton Lowey-Ball, BS, BFA
References: Conti, V., Corbi, G., Sabbatino, F., De Pascale, D., Sellitto, C., Stefanelli, B., … & Filippelli, A. (2023). Long COVID: clinical framing, biomarkers, and therapeutic approaches. Journal of Personalized Medicine, 13(2), 334. https://www.mdpi.com/2075-4426/13/2/334 Koc, H. C., Xiao, J., Liu, W., Li, Y., & Chen, G. (2022). Long COVID and its Management. International Journal of Biological Sciences, 18(12), 4768. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9305273/ Raveendran, A. V., Jayadevan, R., & Sashidharan, S. (2021). Long COVID: an overview. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 15(3), 869-875. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8056514/ Su, Y., Yuan, D., Chen, D. G., Ng, R. H., Wang, K., Choi, J., … & Heath, J. R. (2022). Multiple early factors anticipate post-acute COVID-19 sequelae. Cell, 185(5), 881-895. https://www.sciencedirect.com/science/article/pii/S0092867422000721 Thaweethai, T., Jolley, S. E., Karlson, E. W., Levitan, E. B., Levy, B., McComsey, G. A., … & Donohue, S. E. (2023). Development of a definition of postacute sequelae of SARS-CoV-2 infection. Jama. https://jamanetwork.com/journals/jama/fullarticle/2805540
Scroll down to listen to this article.
Listen to the article here:
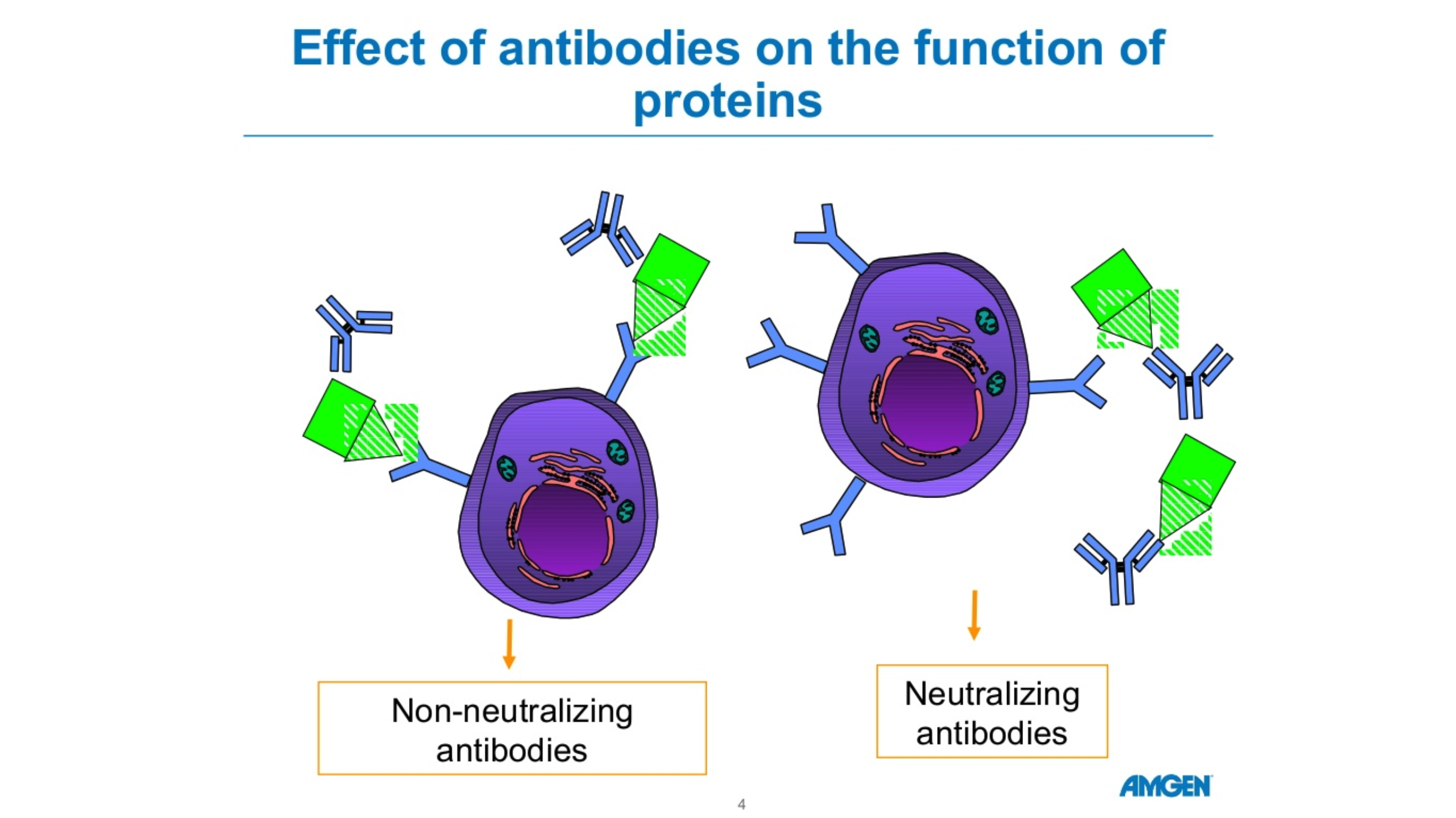
There are five forms of antibodies that the human body makes. There are two forms that are relevant for COVID 19, Igm and IgG. Igm is a big molecule, which is the first molecule that your body makes when you are exposed to a particular antigen or virus. This is an acute phase type of antibody. IgG is a long-term antibody that has memory for your immune system and also protects you long-term. The actual length of long-term protection is not known. Typically, when you have antibody testing, you are tested for both Igm and IgG. These tests are not perfect. If someone tests positive for Igm but not IgG, we’re not sure if they are protected. If someone has no Igm antibodies and lots of IgG antibodies, they’re likely protected due to the long-term memory of IgG. The length of time the antibodies remain detectable following an infection is not known. Source: cdc.gov Amgen Powerpoint