Nature Coast Clinical Research Announces the Remodeling of Its Inverness, Florida Office, Now Offering Phase 1 Clinical Trial Capabilities
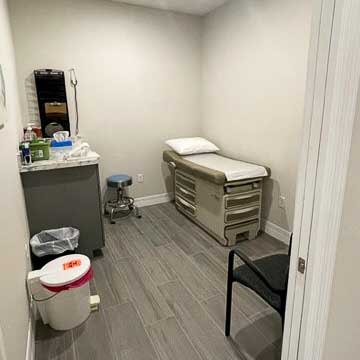
Inverness, Florida – Nature Coast Clinical Research is pleased to announce the completion of our remodeled office in Inverness, Florida, across the street from Citrus HCA Hospital. The fully renovated facility, located at 411 West Highland Boulevard, features new, state-of-the-art equipment and trained staff that allows for an expanded selection of clinical trials.
The remodeling project was undertaken to expand and modernize our research capabilities providing the opportunity for members of the community to participate in all phases of clinical trials. Nature Coast Clinical Research now features a dedicated Phase 1 unit, which includes private inpatient rooms for overnight stays, on-site lifesaving medication and equipment, and certified, trained staff in-house 24/7. The office also has multiple newly renovated, cutting-edge outpatient examination rooms for later-phase trials and non-overnight office visits.
Phase 1, or early phase clinical trials, are used to test the safety of new investigational drugs or devices by helping to determine the correct dosage of new investigational treatments. They are a critical step in the clinical trial process and often require that patients complete overnight stays in the unit.
Phase 2-4, or later phase clinical trials, are conducted on investigational medications or devices that have successfully completed earlier phase trials. They further look at safety parameters as well as dosing regimens, and the effectiveness of investigational therapies and devices.
Nature Coast Clinical Research is part of ENCORE Research Group, a full-service clinical research organization with locations throughout the state of Florida specializing in conducting Phase 1-4 clinical trials for Alzheimer’s, digestive issues, fatty liver, cardiovascular diseases, vaccines, and more. We are committed to providing patients in the community access to the latest treatments and therapies through clinical research studies.
Dr. Rafik Abadier, the medical director of Nature Coast Clinical Research’s Inverness Phase 1 unit, is board-certified and has been practicing cardiology for more than twenty-five years. He has been a Principal Investigator of multiple clinical trials since 2007.
Dr. Paul Hellstern, the medical director of Nature Coast Clinical Research’s Inverness Outpatient office, is board-certified in internal medicine and gastroenterology. He has been a Principal Investigator of multiple clinical trials since 2000.
The remodeled office is now open for business and is accepting patients for all phases of clinical trials. Watch this space to learn more about Nature Coast Clinical Research and our new Phase 1 options!
Contact Jill Livingston, Phase 1 Unit Manager, for more information.
(352) 341-2100

Nature Coast Clinical Research – Inverness
411 West Highland Boulevard Inverness, FL 34452
Main Office: (352) 341-2100